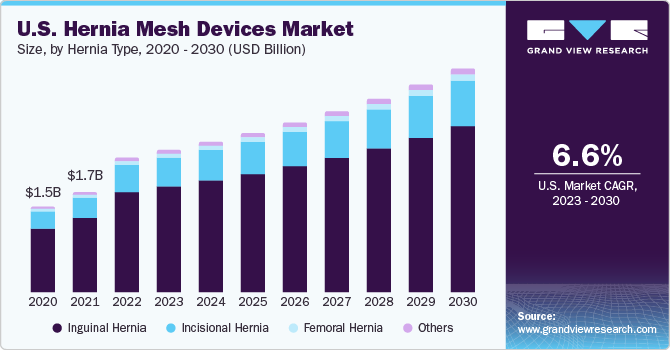
The global hernia mesh devices market is expected to reach USD 199.6
million by 2025, based on a new report by Grand View Research, Inc. The market
is expected to grow over the forecast period owing to increase in incidence of
hernia, high adoption of meshes in surgeries due to factors such as
cost-effectiveness, reduced pain, reduced operative time, and high availability
of products in the market.
The reimbursement scenario for hernia mesh is favorable, which
encourages patients to opt for open or laparoscopic surgery. Patients have easy
access to detailed reimbursement codes and ratios, which are made available by
government and industry players. The codes are granulated to types of hernias,
(primary or recurrent) and method of surgery (open or laparoscopic).
Furthermore, technological advancements and new products introduction,
such as TELA Bio, are driving the overall market growth. In 2010, PROCEED
Ventral Patch (PVP) device was launched by Ethicon for the treatment of umbilical
hernia. In 2016, GORE SYNECOR biomaterial, a product of W. L. Gore &
Associates, was approved by fda as a repair solution for ventral hernia.
Full
Research Report On Hernia Mesh Devices Market Analysis:
http://www.grandviewresearch.com/industry-analysis/hernia-mesh-devices-market
http://www.grandviewresearch.com/industry-analysis/hernia-mesh-devices-market
Further Key Findings from the Study Suggest:
·
Biological surgical meshes is the fastest
growing segment owing to high price of the products and increased preference
for these products by healthcare professionals and patients
·
In February 2016, an EU-funded Green Nano
Mesh was launched for bio-based, eco-friendly treatment of hernia that would
cause less harm to patients
·
Geographically, North America held the
largest market share in 2016 and is expected to maintain its position during
the forecast period. Factors responsible for the large market share are high
incidence of the target disorder and high adoption of surgical meshes as a
treatment option
·
Asia Pacific is also expected to witness
rapid growth over the forecast period. Growing awareness about the target
disorder and improved access to healthcare drives the growth in this region
·
The 13th International Congress of Asia Pacific
Hernia Society is intended to be held in China in September 2017
·
Some of the key players in the hernia mesh
market are Medtronic; Ethicon, Inc., C. R. Bard, Inc.; Atrium Medical; and B.
Braun Melsungen AG.
View More Reports
Of This Category By Grand View Research At: https://www.grandviewresearch.com/industry/healthcare
Grand View Research has segmented the hernia mesh devices market by
hernia type, mesh type and region:
Hernia Type Outlook (Revenue,
USD Million; 2014 - 2025)
·
Inguinal Hernia
·
Incisional Hernia
·
Femoral Hernia
·
Others
Mesh Type Outlook (Market
Revenue in USD Million, 2014 - 2025)
·
Biologic Mesh
·
Synthetic Mesh
Regional Outlook (Market
Revenue in USD Million, 2014 - 2025)
·
North America
o U.S.
o Canada
·
Europe
o UK
o Germany
o France
·
Asia Pacific
o Japan
o China
o India
·
Latin America
o Brazil
o Mexico
·
Middle East and Africa
o South Africa
Access Full Press Release of this
Report:
https://www.grandviewresearch.com/press-release/global-hernia-mesh-devices-market
https://www.grandviewresearch.com/press-release/global-hernia-mesh-devices-market
About Grand View
Research
Grand View Research, Inc. is a U.S.
based market research and consulting company, registered in the State of
California and headquartered in San Francisco. The company provides syndicated
research reports, customized research reports, and consulting services. To help
clients make informed business decisions, we offer market intelligence studies
ensuring relevant and fact-based research across a range of industries, from
technology to chemicals, materials and healthcare.
For more information: https://www.grandviewresearch.com/